High-Fidelity Visibility Into Patient Journeys Can Help CAR-T Cell Therapy Reach More Acute Lymphoblastic Leukemia Patients

This analysis was selected for a poster presentation at ISPOR 2024.
High-fidelity data helps providers identify CAR -T candidates and minimize time to treatment, preventing those who may benefit from falling through the cracks.
For people with acute lymphocytic leukemia (ALL), a typically fast-moving cancer of the blood and bone marrow, timing is crucial. Patients with ALL benefit from swift treatment and a well-facilitated patient journey with no unnecessary delays.
The majority of ALL patients are children, but adults with ALL have worse long-term outcomes overall — they are less likely to respond to treatment and tend to have more complications. Roughly 20% have a primary treatment-resistant disease, and the majority of those who do respond to initial treatments will eventually relapse. The American Society of Clinical Oncology (ASCO) reports that the five-year survival rate for ALL patients age 20 and older is 43% compared to 90% for younger patients. According to a UK-based study, this rate may drop to 7% at five years post-relapse.
A new treatment may change those statistics, however. For those with B-cell ALL whose cancer doesn’t respond to initial treatments, CAR-T cell therapy — a revolutionary emerging treatment — is bringing new hope to some patients. Early research on long-term outcomes of CAR-T for B-cell ALL patients shows promising improvement, especially for children and young adults. However, more time is needed to understand long-term outcomes.
B-cell ALL patients become eligible for CAR-T treatment after one or more of their initial treatments have failed and if they meet other criteria, such as adequate overall health and organ function. A large-scale mapping of patient journeys prior to receiving CAR-T, including common treatments and treatment delays, would give providers more insight into the milestones that patients experience. Our latest analysis investigates the journeys of adult ALL patients who received CAR-T cell therapy, and our hope is that the insights will help inform Life Sciences teams who may be identifying those patients.
Here’s what we found:
Less than 2% of adults who received an initial ALL treatment went on to receive CAR-T cell therapy.
Using data from 2018 to 2021, we identified adult patients diagnosed with ALL who had received at least one form of cancer treatment in the two years following their diagnosis. Of those, 1.8% went on to receive CAR-T cell therapy within two years of that initial treatment.
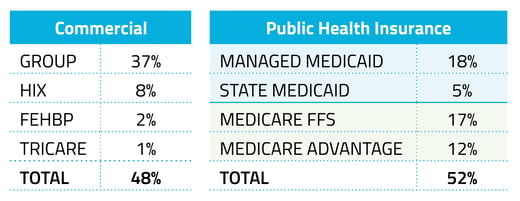 CAR-T cell therapy was more likely to be utilized by patients on commercial insurance than those on Medicare or Medicaid.
CAR-T cell therapy was more likely to be utilized by patients on commercial insurance than those on Medicare or Medicaid.
Just 29% of ALL patients were commercially insured, but this group accounted for 48% of CAR-T recipients. Those on public health insurance made up 71% of ALL patients seen in our Healthcare Map™ but only 52% of those who had received CAR-T cell therapy.
Nearly 1 in 4 ALL patients who receive CAR T-cell therapy has already received at least one bone marrow transplant.
Among those who received CAR T-cell therapy, 38% had received immunotherapy in the two years prior and 21% had received a bone marrow transplant. In the two years prior to their CAR-T treatment, 92% of patients had received chemotherapy or a targeted cancer drug.
 More than half (62%) of patients received CAR-T cell therapy within the month following their last cancer treatment.
More than half (62%) of patients received CAR-T cell therapy within the month following their last cancer treatment.
While this short transition time is encouraging, 14% waited one to two months after their last cancer treatment, and 24% waited two months or more. These longer windows exceed the ideal 15-to-21-day time delay estimate of certain clinics to determine patient eligibility and insurance approval prior to initiating treatment. However, insurance approval can take up to a month, and some patients may continue to receive chemotherapy in the interim.
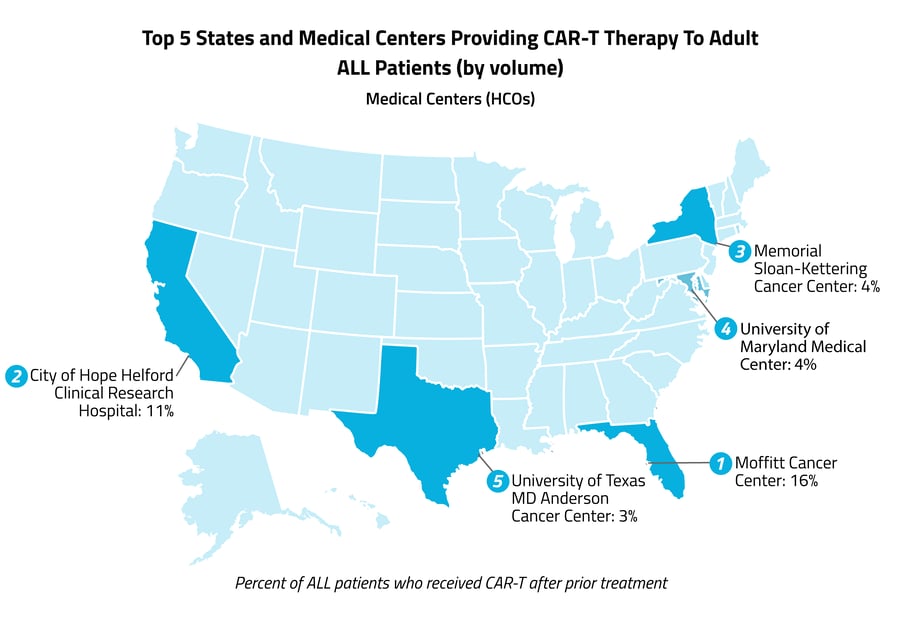
Our findings highlight an untapped potential in both the utilization and efficiency in the utilization of CAR-T cell therapy. As it’s estimated that about 20% of patients with ALL have an initially treatment-resistant form of the disease, there is excellent potential for greater uptake, especially among publicly insured patients. By understanding the journeys of patients undergoing emerging treatments like CAR-T cell therapy, costly pitfalls can be anticipated and sidestepped, eligible patients can be effectively and proactively targeted for treatment, and providers and insurers can have the information they need to make the treatment changeover seamless for patients. This will become increasingly important as CAR-T cell therapy is approved for treating more conditions and the results of ongoing clinical trials are incorporated into new guidelines.
Multiple factors affect whether an ALL patient will receive timely CAR-T cell therapy — eligibility is only one determinant. Provider awareness, access to treatment, insurance approvals, and other delays can play roles. The intensive nature of CAR-T therapy presents its difficulties, as patients must be closely monitored for at least a month after receiving treatment, undergo frequent lab work and scans, and spend at least two weeks in the hospital or make daily outpatient visits. Side effects can also be significant and require additional treatment. For those who do receive CAR-T treatment, the systemic journey often can be unnecessarily slow and expensive. Payers and providers alike will benefit from an ability to anticipate, facilitate, and support these patient transitions.
Our understanding of the risks, benefits, and long-term outcomes of new therapies like CAR-T is still developing, so it is all the more important that experts have access to high-fidelity real-world snapshots of the current state of affairs. Komodo Health integrates patient data with AI and machine learning technologies to offer accurate and up-to-date views of novel and emerging therapies. Reducing the burden of disease, Komodo’s central mission, will require both reactive and proactive measures and high-fidelity data and software to inform and determine the approaches that have the greatest impact.
Read more about our novel, clinically informed computational approaches to capturing the hardest-to-identify patients.
To see more articles like this, follow Komodo Health on X, LinkedIn, or YouTube, and visit Insights on our website.







